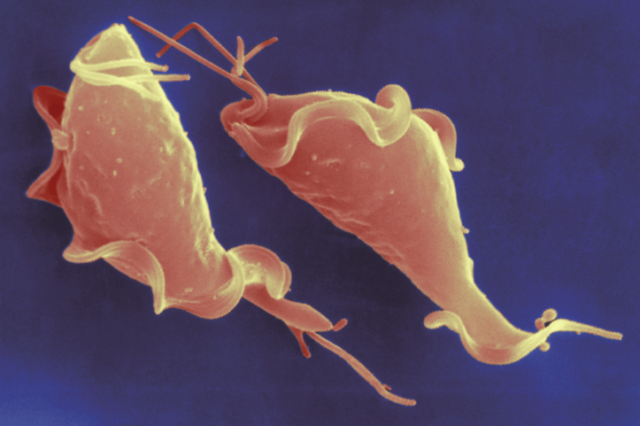
Trichomonas vaginalis is not a very well recognized name – yet, this parasite is responsible for the most common non-viral sexually transmitted infection globally. Human trichomoniasis, the condition caused by the parasitic organism T. vaginalis, is more common than gonnorhea, syphilis, and chlamydia combined, and has been implicated in numerous negative outcomes, including an increased risk of HIV transmission and complications in pregnancy and labor. At the Jane Carlton lab at NYU, I have been able to spend the last 2.5 years working on a team of researchers who seek to better understand T. vaginalis’ evolution and genome.

This summer, I am turning my focus to one T. vaginalis protein in particular: argonaute. A previous graduate researcher at the Carlton lab, Sally Warring, wrote her PhD thesis elucidating the ways in which the genome of T. vaginalis is regulated – that is to say, how certain genes may be silenced, preventing whatever the gene may code for from being formed, or how some genes may be upregulated, increasing production down the line. One mechanism of gene expression that Warring identified is known as RNA interference (RNAi), an ancestral pathway that has been found in many other organisms, in which small RNAs form complexes with specific proteins, notably the argonaute protein, to regulate gene expression. However, experimental methods used in analyzing the argonaute protein have encountered setbacks, as our existing protocols have not yet been optimized for use in the organism T. vaginalis. As such, my research seeks to build upon Warring’s findings by refining our protocols for two common cellular biology experiments in order to understand more about arognaute.
I am just returning to the lab today, where I will work for the next six weeks on these protocols – namely, experiments known as a western blot and immunofluorescence (IFA). The last two weeks I have been preparing for my work in the lab reading protocols and papers in order to understand what we know about argonaute already as well as to observe how other researchers have refined these experiments in similar organisms.
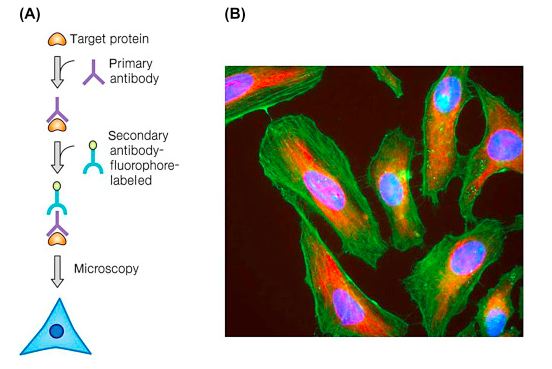
An immunofluorescence experiment involves fixing our cells to a slide, and then incubating the slide with fluorescently labelled antibodies which adhere to our protein of interest. As a result, not only do we get a pretty photo as exemplified below, but we can also visualize the subcellular localization of argonaute, which may suggest the way the protein functions.
Meanwhile, the western blot experiment will allow us to separate the argonaute protein and detect it in samples through a complicated series of steps. While the protocol is daunting, having a refined western blot protocol will be very helpful for broader analysis in the future. The beginning of this week I will be getting settled back into lab, getting organized and growing our cultures. Then I will begin my first immunofluorescence experiment on Thursday, and my first western blot on the following day. Overall, I am excited to be back in lab and report on the preliminary results of my experiments in a few more weeks!